When you first meet her, you won’t be able to tell that Ipek Kuzu suffers from a rare genetic disease. The three-year-old plays happily on her own for hours, driving her toy cars and “cooking” in her pretend kitchen. But she’s not well. She’s a little wobbly on her feet and doesn’t say much, and if nothing is done, she may die by her mid-20s. Ipek has ataxia-telangiectasia, or A-T, a disease caused by an error in her DNA. It causes the loss of brain cells, along with a high risk of infection and cancer.
It’s the sort of problem that makes doctors shake their heads. But Ipek’s father, Mehmet, and mother, Tugba, hope she’ll escape that fate. Thanks in part to the persistence of Mehmet, a programmer at Google, in January she became one of the first handful of US patients to receive a hyper-personalized gene medicine, tailored to treat a unique mutation. The one-person drug, designed for her by a Boston doctor, Timothy Yu, is being called “atipeksen,” for “A-T” and “Ipek.”
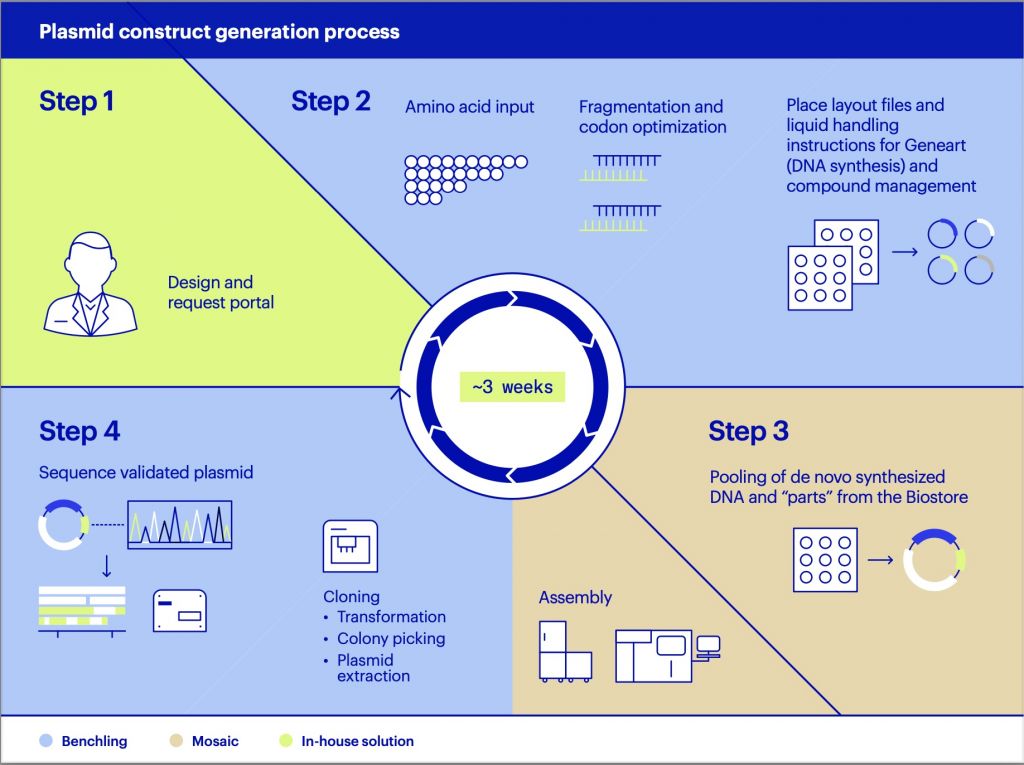
To create atipeksen, Yu borrowed from recent biotech successes like gene therapy. Some new drugs, including cancer therapies, treat disease by directly manipulating genetic information inside a patient’s cells. Now doctors like Yu find they can alter those treatments as if they were digital programs. Change the code, reprogram the drug, and there’s a chance of treating many genetic diseases, even those as unusual as Ipek’s.
The new strategy could in theory help millions of people living with rare diseases, the vast majority of which are caused by genetic typos and have no treatment. US regulators say last year they fielded more than 80 requests to allow genetic treatments for individuals or very small groups, and that they may take steps to make tailor-made medicines easier to try. New technologies, including custom gene-editing treatments using CRISPR, are coming next.
“I never thought we would be in a position to even contemplate trying to help these patients,” says Stanley Crooke, a biotechnology entrepreneur and founder of Ionis Pharmaceuticals, based in Carlsbad, California. “It’s an astonishing moment.”
Antisense drug
Right now, though, insurance companies won’t pay for individualized gene drugs, and no company is making them (though some plan to). Only a few patients have ever gotten them, usually after heroic feats of arm-twisting and fundraising. And it’s no mistake that programmers like Mehmet Kuzu, who works on data privacy, are among the first to pursue individualized drugs. “As computer scientists, they get it. This is all code,” says Ethan Perlstein, chief scientific officer at the Christopher and Dana Reeve Foundation.
A nonprofit, the A-T Children’s Project, funded most of the cost of designing and making Ipek’s drug. For Brad Margus, who created the foundation in 1993 after his two sons were diagnosed with A-T, the change between then and now couldn’t be more dramatic. “We’ve raised so much money, we’ve funded so much research, but it’s so frustrating that the biology just kept getting more and more complex,” he says. “Now, we’re suddenly presented with this opportunity to just fix the problem at its source.”
Ipek was only a few months old when her father began looking for a cure. A geneticist friend sent him a paper describing a possible treatment for her exact form of A-T, and Kuzu flew from Sunnyvale, California, to Los Angeles to meet the scientists behind the research. But they said no one had tried the drug in people: “We need many more years to make this happen,” they told him.
Courtesy Photo (Yu)
Kuzu didn’t have years. After he returned from Los Angeles, Margus handed him a thumb drive with a video of a talk by Yu, a doctor at Boston Children’s Hospital, who described how he planned to treat a young girl with Batten disease (a different neurodegenerative condition) in what press reports would later dub “a stunning illustration of personalized genomic medicine.” Kuzu realized Yu was using the very same gene technology the Los Angeles scientists had dismissed as a pipe dream.
That technology is called “antisense.” Inside a cell, DNA encodes information to make proteins. Between the DNA and the protein, though, come messenger molecules called RNA that ferry the gene information out of the nucleus. Think of antisense as mirror-image molecules that stick to specific RNA messages, letter for letter, blocking them from being made into proteins. It’s possible to silence a gene this way, and sometimes to overcome errors, too.
Though the first antisense drugs appeared 20 years ago, the concept achieved its first blockbuster success only in 2016. That’s when a drug called nusinersen, made by Ionis, was approved to treat children with spinal muscular atrophy, a genetic disease that would otherwise kill them by their second birthday.
Yu, a specialist in gene sequencing, had not worked with antisense before, but once he’d identified the genetic error causing Batten disease in his young patient, Mila Makovec, it became apparent to him he didn’t have to stop there. If he knew the gene error, why not create a gene drug? “All of a sudden a lightbulb went off,” Yu says. “Couldn’t one try to reverse this? It was such an appealing idea, and such a simple idea, that we basically just found ourselves unable to let that go.”
Yu admits it was bold to suggest his idea to Mila’s mother, Julia Vitarello. But he was not starting from scratch. In a demonstration of how modular biotech drugs may become, he based milasen on the same chemistry backbone as the Ionis drug, except he made Mila’s particular mutation the genetic target. Where it had taken decades for Ionis to perfect a drug, Yu now set a record: it took only eight months for him to make milasen, try it on animals, and convince the US Food and Drug Administration to let him inject it into Mila’s spine.
“What’s different now is that someone like Tim Yu can develop a drug with no prior familiarity with this technology,” says Art Krieg, chief scientific officer at Checkmate Pharmaceuticals, based in Cambridge, Massachusetts.
Source code
As word got out about milasen, Yu heard from more than a hundred families asking for his help. That’s put the Boston doctor in a tough position. Yu has plans to try antisense to treat a dozen kids with different diseases, but he knows it’s not the right approach for everyone, and he’s still learning which diseases might be most amenable. And nothing is ever simple—or cheap. Each new version of a drug can behave differently and requires costly safety tests in animals.
Kuzu had the advantage that the Los Angeles researchers had already shown antisense might work. What’s more, Margus agreed that the A-T Children’s Project would help fund the research. But it wouldn’t be fair to make the treatment just for Ipek if the foundation was paying for it. So Margus and Yu decided to test antisense drugs in the cells of three young A-T patients, including Ipek. Whichever kid’s cells responded best would get picked.
Matthew Monteith
While he waited for the test results, Kuzu raised about $200,000 from friends and coworkers at Google. One day, an email landed in his in-box from another Google employee who was fundraising to help a sick child. As he read it, Kuzu felt a jolt of recognition: his coworker, Jennifer Seth, was also working with Yu.
Seth’s daughter Lydia was born in December 2018. The baby, with beautiful chubby cheeks, carries a mutation that causes seizures and may lead to severe disabilities. Seth’s husband Rohan, a well-connected Silicon Valley entrepreneur, refers to the problem as a “tiny random mutation” in her “source code.” The Seths have raised more than $2 million, much of it from co-workers.
Custom drug
By then, Yu was ready to give Kuzu the good news: Ipek’s cells had responded the best. So last September the family packed up and moved from California to Cambridge, Massachusetts, so Ipek could start getting atipeksen. The toddler got her first dose this January, under general anesthesia, through a lumbar puncture into her spine.
After a year, the Kuzus hope to learn whether or not the drug is helping. Doctors will track her brain volume and measure biomarkers in Ipek’s cerebrospinal fluid as a readout of how her disease is progressing. And a team at Johns Hopkins will help compare her movements with those of other kids, both with and without A-T, to observe whether the expected disease symptoms are delayed.
One serious challenge facing gene drugs for individuals is that short of a healing miracle, it may ultimately be impossible to be sure they really work. That’s because the speed with which diseases like A-T progress can vary widely from person to person. Proving a drug is effective, or revealing that it’s a dud, almost always requires collecting data from many patients, not just one. “It’s important for parents who are ready to pay anything, try anything, to appreciate that experimental treatments often don’t work,” says Holly Fernandez Lynch, a lawyer and ethicist at the University of Pennsylvania. “There are risks. Trying one could foreclose other options and even hasten death.”
Kuzu says his family weighed the risks and benefits. “Since this is the first time for this kind of drug, we were a little scared,” he says. But, he concluded, “there’s nothing else to do. This is the only thing that might give hope to us and the other families.”
Another obstacle to ultra-personal drugs is that insurance won’t pay for them. And so far, pharmaceutical companies aren’t interested either. They prioritize drugs that can be sold thousands of times, but as far as anyone knows, Ipek is the only person alive with her exact mutation. That leaves families facing extraordinary financial demands that only the wealthy, lucky, or well connected can meet. Developing Ipek’s treatment has already cost $1.9 million, Margus estimates.
Some scientists think agencies such as the US National Institutes of Health should help fund the research, and will press their case at a meeting in Bethesda, Maryland, in April. Help could also come from the Food and Drug Administration, which is developing guidelines that may speed the work of doctors like Yu. The agency will receive updates on Mila and other patients if any of them experience severe side effects.
The FDA is also considering giving doctors more leeway to modify genetic drugs to try in new patients without securing new permissions each time. Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, likens traditional drug manufacturing to factories that mass-produce identical T-shirts. But, he points out, it’s now possible to order an individual basic T-shirt embroidered with a company logo. So drug manufacturing could become more customized too, Marks believes.
Custom drugs carrying exactly the message a sick kid’s body needs? If we get there, credit will go to companies like Ionis that developed the new types of gene medicine. But it should also go to the Kuzus—and to Brad Margus, Rohan Seth, Julia Vitarello, and all the other parents who are trying save their kids. In doing so, they are turning hyper-personalized medicine into reality.
Erika Check Hayden is director of the science communication program at the University of California, Santa Cruz.